
Recruitment and Apprenticeship Program Update
Orthoplastics experienced significant growth in 2023. Over the last 12 months, we’ve increased our headcount by more than 16%. This growth has been driven by market demand, which is set to remain for the foreseeable future.
We had a fantastic year with our apprentices in 2023; they are now in their first year of college and are progressing well. We expect them back on site in the summer of 2024. We also have four apprentices in their final year with us who will complete the programme in the coming months.
Our apprenticeship intake for 2024
For 2024, our apprentice program is looking to intake another four apprentices. We had a total of 20 direct applicants either from our website, via word of mouth or through the open event we held at Burnley College.
We’ve initially shortlisted these to 14 candidates, and the first round of interviews for these has already begun, which will help us begin the process of shortlisting them further. The stage one interview allows us to meet the applicants, get to know them a little better and find out why they want to be an Orthoplastics apprentice.
Those who are successful will be invited to spend half a day onsite to understand the program better and determine if it’s the right career path for them and if they are a good fit with Orthoplastics. From here, we will offer four applicants a place in our apprenticeship program.
One of our apprentices shared their experiences, stating: “I feel I’m learning new things all the time, from myself and other people. The theory side of college is helping my development on the shop floor. I am happy with the progress I have made since starting my apprenticeship.”
Another continued: “College has played a key part in my development as an engineer, and the progress I have made through my apprenticeship shows in the work I undertake. I feel my apprenticeship has helped to better me as an engineer and worker.”
While another added: “I am really enjoying the practical element of the apprenticeship especially the CNC Machining. It's completely different from being sat in an office like I was doing before. I am getting good support from tutors and from Orthoplastics and even though we are in college, I still feel part of the business. I am looking forward to getting on the shop floor and applying my learning, growing the skills I have learnt so far and progressing in my career.”
Speaking about the apprenticeship program, Danny Rhodes, General Manager at Orthoplastics, said: “The apprentices we have on the program are progressing well, and as a company, we are proud of their achievements to date. When the time comes, I believe many of them could have a bright future ahead of them at Orthoplastics.”
We look forward to welcoming the next intake of apprentices in September and continue to support our current apprentices as they progress through the program.

AAOS a Great Success for Orthoplastics
We were thrilled to participate in the San Francisco AAOS Annual Meeting in February 2024, a unique event that brought together 622 exhibitors and over 7385 visitors. The opportunity to connect with our peers and industry experts in such a dynamic setting was truly invaluable.
During the five days, we engaged in numerous positive discussions with our customers, both existing and potential. We had more than 30 meetings, which allowed us to discuss exciting new products for the future and, reinforced our existing business relationships and helped us shape new ones.
Speaking about the event, Mark Allen, Managing Director of Orthoplastics, said: ”Attending the AAOS Annual Meeting this February in San Francisco was an incredibly rewarding experience for us at Orthoplastics. The event was remarkably well-attended, which was amazing to witness.
The AAOS Annual Meeting has proven itself to be an indispensable platform for industry collaboration, innovation and advancement, and our experience this year has been exceptionally positive.
This event underscores the vibrant future of orthopaedics, and we are truly energised by the potential for growth and innovation that lies ahead. The AAOS Annual Meeting champions progress in our industry, and we were thrilled to be a part of it, paving the way for a future filled with possibilities.”
All in all, AAOS 2024 was a fantastic opportunity for Orthoplastics. We look forward to next year's event in San Diego in March 2025.
Our Expertise
Orthoplastics’ unparalleled expertise in orthopaedic materials is a result of our continuous development over the past 30 years. Our vision of becoming the industry’s leading manufacturer of medical-grade plastics has been realised through our unwavering commitment to quality and excellence.
Investing both time and resources into developing its knowledge of medical plastics and advanced manufacturing techniques has enabled Orthoplastics to continually surpass its customers' exacting standards.
By combining UHMWPE, Cross-linked UHMWPE, and Vitamin E UHMWPE, Component Machining, Direct Compression Moulding and Laboratory Services, Orthoplastics offers its customers a complete supply chain solution.

Orthoplastics are Exhibiting at AAOS
We are thrilled to announce that Orthoplastics will be exhibiting at the biggest orthopaedic technology and innovation exhibition, AAOS 2024. We are excited to join some of the biggest names in the orthopaedic world as more than 600 companies come together to showcase their expertise and gain industry insights.
This year's exhibition will take place at the Moscone Convention Center in San Francisco, CA from Feb 12-16 2024. The AAOS Annual Meeting is poised to deliver one of the best and biggest live education experiences in years, the educational program includes sessions, interactive lectures, courses, and workshops focused on the latest breakthroughs in orthopaedics across all specialities.
Orthoplastics will be located at booth 1028 and we are looking forward to networking with peers and having the opportunity to gain fresh insights and perspectives on the latest technologies and developments within the industry.
Speaking about AAOS 2024, Orthoplastics Managing Director Mark Allen said: “AAOS is a big event on the calendar for the Ortho industry. We are delighted to be returning again in 2024, it’s a fantastic opportunity to see the latest innovations first-hand and speak with peers about their experiences and insights.”
Our Expertise
Orthoplastics’ expert knowledge of orthopaedic materials has been developed continuously over 30 years. The vision of becoming the industry’s leading manufacturer of medical-grade plastics has been achieved through its unsurpassed dedication to quality and excellence.
Investing both time and resources into developing its knowledge of medical plastics and advanced manufacturing techniques, has enabled Orthoplastics to continually surpass its customers' exacting standards.
By bringing together UHMWPE, Cross-linked UHMWPE and Vitamin E UHMWPE, Component Machining, Direct Compression Moulding and Laboratory Services, Orthoplastics offers a complete supply chain solution to its customers.

Championing Engineers of the Future at Orthoplastics
For the last 10 years, Orthoplastics has successfully supported and nurtured the engineering future with its apprenticeship program. Over that decade, 21 apprentices have successfully completed an apprenticeship with and over 70% remain working with the company today.
The Orthoplastics apprenticeship program is offered to candidates through our partnership with Burnley College, a leading college not only in the North of England but across the whole country.
Our apprenticeship program currently takes the apprentice through the first year of off-the-job training, which is fully supported by Burnley College. Apprentices come back on-site during annual leave from the college, before beginning their full on-site training in year 2.
Once back on site in years 2, 3 and 4 they begin a full training program where they have the opportunity to learn all aspects of component machining, including how to program the machines, develop their engineering drawing skills and hone their expertise on our product offering.
The Orthoplastics apprenticeship program 2023
The minimum entry requirement for our apprenticeship program is students who are on target to receive at least a GCSE grade of 4 and above in four subjects including Maths, English and Science.
Successful applicants are invited to attend the first stage interview where they have the opportunity to take a tour of the facilities. During stage 1 we conducted a standard interview to help us find out more about the applicants, why they want to join Orthoplastics and their ambitions for the future.
From the stage 1 interviews, a shortlist of apprentices is created, with a view to making a conditional offer to become an Orthoplastics apprentice. To help us recruit the right candidates for our apprenticeship program, we invited the shortlisted candidates to attend a half-day session with the shift supervisor in the machine shop to give them a better feel for what they will be doing throughout the apprenticeship and to help them decide if engineering is the right career for them.
From this stage, we will shortlist the candidates further and invite the successful applicants to a more in-depth tour of the facilities and further conversations to ensure that there is a good fit between the candidate and Orthoplastics.
We are pleased to share that we have now concluded our Apprenticeship intake for 2023 where we have made six conditional offers, all of which have been accepted.
Orthoplastics apprenticeships success stories
One of our success stories from the apprenticeship program is an individual that joined the company as an apprentice in 2017, and through hard work, dedication and support from Orthoplastics has experienced a quick progression through the business and is now part of the new product introduction team.
We are also incredibly proud that one of our female apprentices was awarded Apprentice of the Year 2023 by Burnley College. She is about to progress to her second year of the program and has a bright future in engineering ahead of her.

Speaking about the apprenticeship program, Danny Rhodes, General Manager at Orthoplastics said: “We are proud to support the local community with our apprenticeship program and provide unique opportunities for young adults. Our program enables us to work with and shape the apprentices into becoming the future engineers of Orthoplastics.
There are many avenues for the apprentices to progress into post their apprenticeship and we will continue to work with the apprentice to invest and support their future within the company.“
We look forward to welcoming the next intake of apprentices in September and continue to support our current apprentices as they progress through the program.

Come See Us at OMTEC 2023
We are excited to announce that Orthoplastics will be exhibiting at OMTEC 2023. This year's annual Orthopaedic Manufacturing & Technology Exposition and Conference (OMTEC) will be held at the Donald E. Stephens Convention Center in Chicago on 13-15 June 2023.
We will be joining a range of orthopaedic professionals and organisations including OEMs, suppliers, consultants, surgeons, societies, regulatory bodies, and universities. Orthoplastics will be located on booth 111 and we are looking forward to showcasing our world-leading products and laboratory services.
Speaking about OMTEC 2023, Orthoplastics Managing Director Mark Allen said: “OMTEC is a one-of-a-kind event that brings leaders in the global orthopaedic manufacturing industry together to share their insights, perspectives and discover the latest innovations. We are excited to be exhibiting again this year and look forward to connecting with our peers.”
The 3-day event will be packed with education sessions, networking events and 160 orthopaedic-focused suppliers and service providers covering essential capabilities such as implant and instrument manufacturing, 3D printing, surface technologies and regulatory affairs.
Our Expertise
Orthoplastics’ expert knowledge of orthopaedic materials has been developed continuously over 30 years. The vision of becoming the industry’s leading manufacturer of medical-grade plastics has been achieved through its unsurpassed dedication to quality and excellence.
Investing both time and resources into developing its knowledge of medical plastics and advanced manufacturing techniques, has enabled Orthoplastics to continually surpass its customers' exacting standards.
By bringing together UHMWPE, Cross-linked UHMWPE and Vitamin E UHMWPE, Component Machining, Direct Compression Moulding and Laboratory Services, Orthoplastics offers a complete supply chain solution to its customers.
We look forward to meeting new prospects and connecting with like-minded peers across the orthopaedic manufacturing community.

Orthoplastics Welcomes Our New General Manager
We are pleased to announce that Orthoplastics has recently appointed a new General Manager, Danny Rhodes. Danny joined the team in January 2023, following an impressive management career beginning with 17 years at Rolls Royce.
Danny began working for Rolls Royce after securing an apprenticeship upon leaving school and then worked his way towards his first management role in 2015. Prior to joining Orthoplastics, Danny was the Head of Manufacturing for an engineering company specialising in the aerospace and power markets.
Bringing the skill set he has built up over his career, Danny’s role as General Manager at Orthoplastics is to oversee operations and help coach other managers within the company.
Speaking on the role, Danny says: “Orthoplastics is already a strong business with excellent market penetration, but my aim is to help the business go from strength to strength by enhancing the management team.”

Having worked in both a blue chip company and an SME, Danny has an exceptional understanding of how both types of companies operate and how the strategies can be blended and carried forward at Orthoplastics to secure its growth and resilience, now and in the future.
Danny hopes to bring in elements from his MBA course to help him implement strategic planning such as digitisation and project planning to help facilitate business growth and the transition to a larger company.
When not working, father of two Danny enjoys spending time with his wife and young family. Whether that be taking the family dog on a long walk or enjoying a well-earned weekend break in their caravan.
We’d like to take this opportunity to welcome Danny to the team and we look forward to an exciting future of working together to grow and further enhance Orthoplastics.

Orthoplastics are Exhibiting at AAOS in March
We are pleased to announce that Orthoplastics will be exhibiting at the biggest orthopaedic technology and innovation exhibition, AAOS 2023. We are excited to join some of the biggest names in the orthopaedic world as more than 600 companies come together to showcase their expertise and gain industry insights.
This year's exhibition will be held at the Venetian Convention & Expo Centre in Las Vegas, Nevada from March 7-11 2024. The AAOS 2023 Annual Meeting is poised to deliver one of the best and biggest live education experiences in years, the educational program includes sessions, interactive lectures, courses, and workshops focused on the latest breakthroughs in orthopaedics across all specialities.
Orthoplastics will be located in Hall A Booth 1544 and we are looking forward to networking with peers and having the opportunity to gain fresh insights and perspectives on the latest technologies and developments within the industry.
Speaking about AAOS 2023, Orthoplastics Managing Director Mark Allen said: “As the end of the first quarter of another exciting year draws closer, we are looking forward to exhibiting at one of the biggest orthopaedic events in the world. It is a great opportunity to see the latest innovations first-hand and speak with peers about their experiences and insights.”
Our Expertise
Orthoplastics’ expert knowledge of orthopaedic materials has been developed continuously over 30 years. The vision of becoming the industry’s leading manufacturer of medical grade plastics has been achieved through its unsurpassed dedication to quality and excellence.
Investing both time and resources into developing its knowledge of medical plastics and advanced manufacturing techniques, has enabled Orthoplastics to continually surpass its customers exacting standards.
By bringing together UHMWPE, Cross-linked UHMWPE and Vitamin E UHMWPE, Component Machining, Direct Compression Moulding and Laboratory Services, Orthoplastics offers a complete supply chain solution to its customers.

Orthoplastics (a Viant company) Collects 742lb of Food for Local Food Banks
The current cost of living crisis is making the vital role food banks play in the local community even more crucial and here at Orthoplastics we wanted to do our bit to help two local food banks keep their shelves stocked this winter.
We launched a company-wide food drive from 23rd November to 2nd December to encourage employees to donate any unwanted food items such as tinned meat, soups, vegetables, beans and potatoes, rice, pasta, condiments and canned fruits.
Tote bags were placed at several key places within the premises to make it easy for employees to leave their donations and we are super proud of the efforts everyone has shown.
The results of our food drive have been overwhelmingly positive and will make a significant difference to our two chosen food banks this winter and the people within the local community that
rely on them.


Together, we have collected 742lb of food to be donated to the Trinity Baptist Church and The Hare & Hounds Pub Food Box on the Side, both of which do wonderful work in the local community to help those struggling with the cost of living.
The generosity shown by employees has been amazing and we hope that our donations will go a long way to help these two fantastic food banks over the winter. Well done to everyone who was involved.

Orthoplastics Are Exhibiting at OMTEC
We are pleased to announce that Orthoplastics will be exhibiting at the 16th Annual Orthopaedic Manufacturing & Technology Exposition and Conference (OMTEC) at the Donald E. Stephens Convention Center in Chicago on 14-16 June 2022.
Due to the COVID-19 pandemic, the event last took place in 2019. But this year is set to make a spectacular return with a packed full agenda of executive insight on the state and future of the orthopaedic industry, live demonstrations and discussions around supply chain challenges and how to overcome them.
Recognised as an international conference that facilitates dialogue and partnerships among suppliers, service providers and orthopaedic OEM professionals, OMTEC is the only conference exclusively serving the orthopaedic industry.
With a dedication to the medical sector and as a producer of the highest quality, premium grade orthopaedic UHMWPE on the market, we are delighted to be attending OMTEC. We will be showcasing our world leading products and laboratory services on booth 604.
Orthoplastics bring together UHMWPE, Crosslinked UHMWPE and Vitamin E UHMWPE, Component Machining, Direct Compression Moulding and Laboratory Services to provide the complete supply chain solution to our customers.
We look forward to meeting new prospects and connecting with like-minded peers across the orthopaedic manufacturing community.

New Manufacturing Facility Helping Support the Local Community
In a bid to help the local community access COVID-19 testing and vaccinations, we are proud that our new manufacturing facility could be used as a temporary COVID vaccination and testing centre.
Using our facility as a temporary testing and vaccination centre meant that more people were able to access these services in an easier, more convenient way. Located on Futures Business Park the centre was easy for people to get to and there was plenty of parking available.
Managing Director, Mark Allen said: “We are pleased to be doing our bit in the fight against COVID-19 by allowing Rossendale Council to use our new manufacturing facility. Given the size of the facility it is ideal and enables people to safely social distance while accessing tests or vaccinations.”

Testing and vaccination are our best defence in bringing the virus under control. People were able to have their vaccines without an appointment, and with cases rising in the local area, we were delighted to be able to offer the use of our facility to support the local community.
Clinicians were on hand at the centre to answer any questions and address any concerns people may have. The centre operated 7 days a week from 8am and 8pm, giving people plenty of opportunity to visit.
We would like to thank Rossendale Council and all the volunteers at the testing and vaccine centres for their hard work and dedication to helping minimise the spread of the virus.
New facility set to open end of 2021
The opening of the new facility is expected to happen towards the end of 2021 and once we are ready to begin manufacturing it will enable us to further increase our market capabilities with over £5 million of investment annually.
Our expansion not only increases our capabilities but will create many job opportunities within the local community once open and in the future.

Further Expansion of our Direct Compression Moulding Capabilities
We are pleased to announce that we have further expanded our Direct Compression Moulding capabilities. We took delivery of a batch of DCM machines back in December 2020 and took a further delivery of additional machines in January 2021.
Managing Director, Mark Allen said: “This is great news for Orthoplastics, following the first expansion of our Direct Compression Moulding capabilities at the end of 2019 we made the decision to expand further to help us meet customer demand, advance orthopaedic developments and exceed patient expectations.”
Orthoplastics is focused on advanced methods of manufacturing UHMWPE and is a world leader in Direct Compression Moulding technology. Using our technological expertise, we can bring to market new innovative product solutions to address failure modes typical within orthopaedic implants.
We work in partnership with many OEMS around the world to enhance product quality and improve outcomes for patients. Our knowledge enables us to navigate our partners through the complexities of production, compliance and accreditation, helping to reduce costs and accelerate speed to market.
Leading the World
Our Direct Compression Moulding services are at the cutting edge of new product development. With each component being individually moulded it allows for the finite controlling of all aspects of pressure, temperature and time, during critical consolidation.
On-site Laboratory Services
We work with our customers to accelerate the production process and ensure that all new product concepts meet the application, specification and technical demands required. We have our own on-site research and development laboratory that is extensively equipped and available for use by our customers.
As a World leader in UHMWPE manufacturing, we have over 30 years’ experience of producing orthopaedic components. We bring together UHMWPE, Cross Linked UHMWPE and Vitamin E UHMWE, Machined Components, Direct Compression Moulding and Laboratory Services to provide the complete supply chain solution.
Discover more about our Direct Compression Moulding expertise at directcompressionmoulding.com

Orthoplastics Facilities Team Announced as Winners of the Viant Values 2021 Q1 Awards
The Facilities team worked through Christmas to undertake essential Prepared Planned Maintenance work to ensure the facility was up and running for the return of all employees. Facilities team as pictured above L-R:
Brian Green - Facilities Manager
John Proctor - Facilities Engineer
Lewis Tattersall - Health, Safety & Environmental Officer
Neil Anderton - Business Development Director
David Hamilton - Electrical Maintenance Engineer
They managed power cuts, floods and major equipment failures working out of hours to ensure the safety of our employees. Working incredibly unsociable hours to ensure the facility kept going during the recent catalogue of events. Their attitude was second to none, always willing to get the job done for the good of the company.
The team continue to maintain CV-19 awareness for employees and contractors, ensuring PPE implementation, ensuring non-compliance with CV-19 restrictions is dealt with by management and for a quarter of the year ensured temperature testing for the facility on a 24/7 basis until scanning machines were installed. One of the team has a young family but continued to assist the rest of the Facilities Team without complaint.
In addition to the above, they have all participated in the new facility build for the past 6 months ensuring we are on target.
On behalf of all at Orthoplastics may we say a huge thank you and well done!
Mark Allen
Managing Director

Progress at our New Manufacturing Facility
We’re pleased to update you on the progress at our new manufacturing facility at the Futures Business Park Development in Rossendale (UK). As with all complex builds, the path to completion has not been a smooth one and the added complications of the COVID-19 pandemic has brought its own unique set of challenges.
One of the biggest challenges we have faced over the last 12 months has been contractor availability and continuity of work during lockdown and periods of restriction due to the pandemic. This was a challenge that could not be foreseen when work first began back in 2019, but it was one we have had to navigate.
Despite the setbacks and delays, we’re pleased with the progress we have made at the new building. The hydraulic press is now in-situ, it has been tested and we can confirm it is working, this was a monumental step forward for us as delivery of the press was complex enough without the additional challenges that COVID presented us with.


We are now just waiting for the validation on the press so it can be operational and this is expected to take place in mid-April. We are also only a few weeks away from the inside of the building being completed. The exterior of the building is complete with new signage and the car park being finished.


The opening of the new facility is expected to happen towards the end of 2021 and once we are ready to begin manufacturing it will enable us to further increase our market capabilities with over £5 million of investment annually. Our expansion not only increases our capabilities but will create many job opportunities within the local community once open and in the future.
Business Development Director, Neil Anderton said: “It was impossible to predict the challenges we would face when we first began the project in 2019, but our determination and tenacious approach has got us through and we are proud of the progress we have made and look forward to the opening of the facility.”

Keeping Operations Running While Ensuring the Safety of our People
2020 presented challenges for many businesses, we’re proud that since April 2020 we have put stringent measures in place to ensure our facilities are COVID secure and that the health and safety of our employees is of paramount importance. Since April 2020 we have been able to resume operations in a safe and secure environment.
Following government guidelines on safe working practices during the pandemic, we have made sure that our employees are able to practice social distancing and follow procedures set out to make social distancing achievable.
Access to the necessary Personal Protective Equipment such as gloves and masks has been provided where needed to help keep our teams protected and mitigate risks. We have also ensured that hand cleaning and sanitising facilities are located throughout the workplace and that surfaces and equipment are cleaned more frequently and in line with government guidance.
Orthoplastics Managing Director, Mark Allen said: “Here at Orthoplastics, our priority is always our people. We are doing everything we can to make sure that our employees’ health, safety and wellbeing is at the core of everything we do.
Aside from the changes to the workplace and our working practices, we have also implemented a COVID team who regularly meet to review the latest government advice to make sure we are following the most up to date guidelines and whether there are any further actions required to remain COVID secure. We have kept employees up to date and made sure they are involved in any changes to working practices to ensure the safety measures are adhered to and effective.”
Keeping operations running safely
Orthoplastics are proud to have resumed operations to ensure our customers’ requirements are met while ensuring the safety of our people.
As part of the Viant corporation, Orthoplastics is dedicated to the medical industry and is a world leader in the development and supply of both implantable-grade plastics and also biocompatible plastics for instrument applications.
We manufacture implantable plastics, producing the highest grade orthopaedic UHMWPE and medical devices on the market.
By bringing together UHMWPE, Cross-linked UHMWPE and Vitamin E UHMWPE, Component Machining, Direct Compression Moulding and Laboratory Services, Orthoplastics offers a complete supply chain solution to its customers.
Our premium grade UHMWPE is expertly manufactured onsite in the UK and is the material of choice in a wide range of orthopaedic implants and devices for applications in hips, knees, shoulders, elbows, ankles and fingers.

Orthoplastics: Part of the Viant Corporation
Orthoplastics is dedicated to the medical industry and is a world leader in the development and supply of both implantable-grade plastics and also biocompatible plastics for instrument applications.
We manufacture implantable plastics, producing the highest grade orthopaedic UHMWPE and medical devices on the market. Our product development expertise is trusted and proven, with our implantable plastics used in orthopaedic implant applications such as knees, hips and shoulders.
As a part of the Viant corporation, our long term strategic goals at Orthoplastics are to become a true, end-to-end partner for an efficient supply chain solution.
About Viant
Viant is focussed on providing end-to-end solutions for medical device OEMs. Their deep materials expertise, including metals, combined with our experience in engineering, manufacturing, assembly, packaging, and sterilisation allows us to bring our customers’ medical technology solutions to market.
With over 25 worldwide locations and around 425 precision machining centres, Viant is well-positioned to serve its OEM partners and are at the forefront of orthopaedics. With deep expertise in both plastic and metal materials, they provide complete solutions including:
● Implants: low- to high-volume manufacturing; advanced coating capabilities
● Instruments: traditional and robotic; reusable and single use
● Delivery systems: best-in-class design; low- to high-volume manufacturing
Commitment to quality
At Orthoplastics, quality encompasses everything across all aspects of production, from customer service, to design and manufacture of its premium grade medical plastics.
As part of the Viant corporation, quality is at the forefront of what we do every day because our components and finished devices save or enhance lives every day and help medical professionals achieve better outcomes for patients.
To emphasise this strict focus on delivering the highest grade of UHMWPE products, Orthoplastics adhere to rigorous quality inspection standards and processes. This ensures that every product that leaves its facility is guaranteed to meet and exceed customers demanding standards.
As a result, Orthoplastics have been accredited to a wide range of International Quality Standards. Paying full attention towards the customer and the product ensures that Orthoplastics fully understand the needs of their clients, even down to the packaging and delivery of the product.
Orthoplastics Limited is dedicated to the medical industry and is a world leader in the development and supply of both implantable grade plastics and also biocompatible plastics for instrument applications. Get in touch with us to discuss your requirements and discover how we can help.

Orthoplastics: We’ve Extended our Instrument Materials
Orthoplastics has been a manufacturer of instrument materials since 2002, recently we have invested in new manufacturing capabilities which adhere to the latest international standards. Our investment of time and resources into innovation has created a platform whereby Orthoplastics can continue to improve on its high quality, industry-leading products.
Here at Orthoplastics, we have extended our materials offering with Polyacetal (Hostaform) and Polyphenylsulphone.
Hostaform© is the latest offering from Celanese healthcare, a long term replacement for its legacy Celcon portfolio, this material complies to ASTM F1855 and is available in a wide range of colours to meet the demand for high-quality instrument applications and improve quality in coloured molded parts.
Using Hostaform for coloured parts delivers increased polymer stability due to reduced heat history during the preparation of the coloured polymer, improved colour distribution throughout the injection molding process and reduces risks in medical innovations with stable, high-quality and regulatory compliant colours.
PPSu is a high-performance resin that has been available for use in the medical sector for many years. Orthoplastics recently worked with the supplier to develop the extensive validations for the manufacture of the extruded bar stock. Again, the wide range of colours meets the industry demands for identification, traceability and long-term performance.
Polyphenylsulfone (PPSU) can withstand continuous exposure to moisture and high temperatures and still absorb tremendous impact without cracking or breaking.
All materials are available in standards 5ft (1525mm) lengths and a diameter range from 1”(25.4mm) – 4”(101.6mm)
Industry-leading systems & facilities
Orthoplastics’ industry-leading facilities are situated around 50 kilometres north of the city of Manchester. Its impressive facilities offer a combined size that exceeds 50,000 square feet.
Firstly, Orthoplastics 30,000 square foot unit contains a range of different departments including machining, finishing, research & development, shipping and warehousing space. Additionally, a 20,000 square foot press facility has been purpose built in order to house the state of the art UHMWPE press that has been installed at Orthoplastics and offers an exceptional quality of finish on all products manufactured.
Orthoplastics Limited is dedicated to the medical industry and is a world leader in the development and supply of both implantable grade plastics and also biocompatible plastics for instrument applications. Get in touch with us to discuss your requirements and discover how we can help.

Quality Management System Statement
During the COVID-19 outbreak, Orthoplastics have adapted their current working practices to ensure that all aspects of our accredited QMS and regulatory and compliance activates are adhered to, despite current restrictions in place.
Orthoplastics is fortunate enough to already have an electronic QMS in place, which has made working remotely and ensuring compliance to standards more accessible for our Quality Management team.
Orthoplastics is working closely with accreditation bodies to ensure our quality management systems are maintained and reviewed remotely. We are extremely proud of how we have continued our high-quality standards, as well as our robust Quality Management System during this time.
We are pleased to announce that we have been certified to ISO 17025:2017, which has been achieved by remote based working.
As a medical device supplier, many of our customers require a supplier audit to be performed periodically. As such we have adapted our audit process internally and externally to ensure the safety of our employees, customers and suppliers.
We are now offering our customers desktop and virtual audits of all aspects of our Quality Management System, until at least 2021. To arrange a desktop / virtual audit please contact Rebecca Field by email at field@orthoplastics.com
Despite these uncertain times, Orthoplastics remain dedicated to the continued conformity of all its quality management systems ensuring product quality is at the forefront of all working practices.
Committed to Delivering the Highest Quality Standards
At Orthoplastics, quality is at the core of everything we do and covers all aspects of production, customer service, design and the manufacture of our premium-grade medical plastics.
To emphasise this strict focus on delivering the highest grade of UHMWPE products, Orthoplastics adhere to rigorous quality inspection standards and processes. This ensures that every product that leaves its facility is guaranteed to meet and exceed customers demanding standards.
As a result of this devoted approach towards quality, Orthoplastics have been accredited to a wide range of International Quality Standards.
Employing traceability techniques during the manufacturing of all medical-grade product, allows Orthoplastics to regularly assess product quality. All data is recorded and stored for a minimum of 30 years after the manufacturing processes.
Additionally, implementing visual inspection techniques allows for the determination of the products’ quality, along with using non-destructive, ultrasonic testing. Product batch testing is then completed in accordance with ISO 5834-2, ASTM F648 and customer specifications.

What’s Been Happening at Orthoplastics in 2020
2020 has been a year like no other and we’re only just over halfway through. From torrential rain to a heatwave the weather has certainly been unpredictable and challenging.
The COVID pandemic and lockdown brought challenges that many of us didn’t expect and we all had to learn to adapt quickly, not only professionally but personally too. That said, here at Orthoplastics, we have some exciting news that we are proud to share with you.
New Manufacturing Facility Update
Back in September of last year, we announced plans for the opening of an additional manufacturing facility in the UK and we’re pleased to have an update on the progress of the new building at the Futures Business Park Development in Rossendale.
As with all complex builds, the path to completion never runs smooth and this build hasn’t been an exception, but we’re pleased with how it is progressing.


The new build at Futures Park continues at a pace with the partial completion handover being 10th July for the internals only 2 weeks over schedule, but given the weather and COVID, this is an excellent result.
Whilst the internals will be finished, it will be a further 2 weeks for all external works such as car parks and landscaping to be complete and they should be finished by the end of July 2020.
The building completion is excellent, but we have encountered delays on the press build due to the COVID lockdown and travel restrictions. Approximately 25% of the press build is complete and the remaining build is estimated to take around 10-15 days once the engineers return.
Progress Update on Apprentices
As education has been one of the biggest areas that has been impacted by COVID, it was inevitable that apprenticeships would be affected during these unprecedented times.
We’re pleased to say that colleges such as Burnley College were quick to respond and adapt and offered remote learning for the apprentices that usually attend college for their day release qualification and special measures have been put in place to overcome the challenges of workplace assessments.
Here at Orthoplastics, we have been encouraging our apprentices to continue with their studies during lockdown.

Queen’s Awards Win Opens the Door to New Opportunities for Orthoplastics
During a time of uncertainty and unrest across the country and around the globe, being awarded the Queen’s Awards for Enterprise and International Trade couldn’t have come at a better time to boost staff morale.
The whole team here at Orthoplastics are incredibly proud that their hard work, commitment and determination has been recognised with such a prestigious award.
The awards celebrate the success of exciting and innovative businesses which are leading the way with pioneering products or services, delivering impressive social mobility programmes or showing their commitment to excellent sustainable development practices.
Increased recognition nationally and internationally
Sales and Marketing Director, Bernadette Dabbs says “not only has the Queen’s Award given a great boost to staff morale during these uncertain times, but it also opens the door to new opportunities for Orthoplastics both nationally and internationally.
Recognised around the world as the royal seal of approval, it’s the highest accolade any UK business can achieve and will help us drive forward our international marketing efforts and raise the profile of the business.
Use of the Queen’s Award emblem in our marketing materials will help us reinforce our achievement and gain a competitive edge. It also opens the door to increased press coverage not only at home but abroad too. It’s an exciting time here at Orthoplastics.”
A great boost for the company
Mark Allen, Managing Director at Orthoplastics says “I’m am incredibly proud of the team. Winning this award is testament to the hard work and commitment of everyone here at Orthoplastics. News of the award has really given a boost to morale across the board.
Getting the royal seal of approval confirms that Orthoplastics is a leader in the manufacture of medical-grade plastics for the medical sector and the award will help us gain recognition around the world and open up new opportunities for us as a company.
Everyone here should be proud of the achievement and we are all looking forward to the award presentation.”
Orthoplastics is a fully dedicated end-to-end solution provider to the medical sector and has developed an international presence with over 350 global OEM’s producing implantable orthopaedic products.
Orthoplastics’ expert knowledge of orthopaedic materials has been developed continuously and our vision of becoming the industry’s leading manufacturer of medical-grade plastics has been achieved through our unsurpassed dedication to quality and excellence.

A History of the Queen’s Awards for Enterprise
Following the announcement of our delight at being awarded the prestigious Queen’s Awards for Enterprise and International Trade, we wanted to share with you a history of the Queen’s Awards.
When where the Queen’s Awards established?
The Queen's Awards for Enterprise is an awards programme for British businesses and other organisations who excel at international trade, innovation or sustainable development. It’s the highest official UK award a British business can be awarded.
The scheme was established as The Queen’s Awards to Industry in 1965 and included The Queen's Awards for Export, Export Achievement, Technology, Technological Achievement and Environmental Achievement. Until 2017, when it was rested, the scheme also included an award for individuals known as the Queen’s Award for Enterprise Promotion.
The Queen's Award to Industry became the Queen's Awards for Export and Technology in 1975, with Environmental Achievement added in 1992. In 1999, the scheme became the Queen's Awards for Enterprise with International Trade, Innovation and Sustainable Development as the categories. In 2017, a fourth category was introduced, Promoting Opportunity (through social mobility).
Eligibility to enter the Queen’s Award for Enterprise
To apply for the Queen’s Award for Enterprise your organisation must:
● Your organisation can be a business or non-profit and be based in the UK (including the Channel Islands and the Isle of Man)
● Tile its Company Tax Returns with HM Revenue and Customs (HMRC)
● Be a self-contained enterprise that markets its own products or services and is under its own management
● Have at least 2 full-time UK employees or part-time equivalents
● Demonstrate strong corporate social responsibility
The Queen’s Award for Enterprise 2020
Since the awards were first established over 7000 companies have achieved a Queen’s Awards, including Orthoplastics who were awarded the Queen’s Awards for Enterprise and International Trade 2020.
Now in its 54th year, the Queen’s Awards for Enterprise remain the most prestigious business awards in the country, with winning businesses able to use the esteemed Queen’s Awards emblem for a five year period. Bringing with it more recognition, nationally and internationally and greater opportunities.
This year 220 UK businesses have been recognised for their contribution to International Trade, Innovation, Sustainable Development and Promoting Opportunity (through Social Mobility).
The Queen’s Awards for Enterprise 2020 include:
● 128 awards for International Trade
● 66 awards for Innovation
● 19 awards for Sustainable Development
● 7 awards for Promoting Opportunity (through Social Mobility)
What happens if your company wins?
The Queen’s Award is valid for five years and winning organisations will benefit from being able to fly the Queen’s Award flag at their principal premises, use of the emblem on their stationery, advertising and goods and Global recognition and commercial value for your business.
Winning a Queen’s Award for Enterprise will also help your company gain a competitive edge and will open up new business opportunities. In addition, winning the highest UK business award can provide a great boost to staff morale.
Winning businesses will be presented with the award at the winners’ premises by the Lord-Lieutenant of the County as HM The Queen’s representative. The Awards consist of a Grant of Appointment and a presentational item bearing the Emblem. Representatives of the winning businesses will also be invited to a celebratory Royal Reception.
We are extremely proud to be awarded our first Queen’s Awards for Enterprise and International trade. The award was confirmed on Tuesday 21st April and is an outstanding achievement for the company.
Managing Director Mark Allen says “this is a fantastic achievement for the whole team at Orthoplastics and is a true testament to the hard work and dedication of everyone here. Being awarded such a prestigious award is a great morale boost and will open the door to many new opportunities. We’re looking forward to the award presentation and attending the Royal Reception.”
The award is recognition of Orthoplastics being the leading manufacturer of medical-grade plastics, which has been achieved through our unsurpassed dedication to quality and excellence. Investing both time and resources into developing our knowledge of medical plastics and advanced manufacturing techniques has enabled us to continually surpass our customers exacting standards.

Orthoplastics awarded Queen’s Awards for Enterprise and International Trade
We are extremely proud to announce Orthoplastics has been awarded our first Queen’s Awards for Enterprise and International trade.
Orthoplastics is a fully dedicated end-to-end solution provider to the medical sector and has developed an international presence with over 350 global OEM’s producing implantable orthopaedic products.
Orthoplastics’ expert knowledge of orthopaedic materials has been developed continuously and our vision of becoming the industry’s leading manufacturer of medical-grade plastics has been achieved through our unsurpassed dedication to quality and excellence.
Investing both time and resources into developing our knowledge of medical plastics and advanced manufacturing techniques has enabled us to continually surpass our customers exacting standards.
Science making a material difference
Our products were initially pioneered in the UK around 1969 for orthopaedic applications and have over 50 years of successful clinical history throughout the world.
Orthoplastics has been supplying materials for life-changing surgeries since 1996 and has led the way in making recognisable contributions to joint replacement. Our motto ‘science making a material difference’ truly promises an extended product life span and a greatly improved quality of life for patients that receive orthopaedic implants.
In recent years, orthopaedic device manufacturers have identified UHMWPE as the most suitable orthopaedic material to use when creating medical-grade applications for the health sector.
Orthoplastics have drawn upon its industry expertise in medical plastics to provide a series of integrated processes to produce the highest quality of UHMWPE in the world. It’s this premium-grade product that is now the material of choice for orthopaedic device manufacturers across the globe.
Its advanced industry knowledge provides Orthoplastics with the opportunity to continually develop and manufacture innovative product solutions for the medical industry.
An outstanding achievement
We are extremely proud to be awarded our first Queen’s Awards for Enterprise and International trade. The award was confirmed on Tuesday 21st April and is an outstanding achievement for the whole team.
Managing Director Mark Allen says “winning such a prestigious award confirms that Orthoplastics is a leader in the manufacture of medical-grade plastics for the medical sector. The Queen’s Award for Enterprise is the highest accolade for British businesses, bringing with it a host of new opportunities and recognition nationally and internationally.”
The Queen’s Awards for Enterprise are the most prestigious business awards in the UK. The awards were first established in 1965 and since then over 7000 companies have achieved a Queen’s Awards.
The awards celebrate the success of exciting and innovative businesses which are leading the way with pioneering products or services, delivering impressive social mobility programmes or showing their commitment to excellent sustainable development practices.
We are uniquely placed to service global providers with not just materials, but semi-finished devices manufactured in a highly regulated environment using bespoke process techniques. As part of the ‘Viant Inc’ group, our depth of knowledge and capabilities will enhance patients’ lives forever.

COVID19 Update: Partial Operations Resumed
Dear Valued Customer,
Following our recent temporary closure of the facility due to the COVID19 crisis, we would like to provide you with an update
The Orthoplastics facility resumed partial operations as of 14th April.
All functions within the building are operating on limited resources to protect our employees and adhere to the latest government guidance. Obviously during this time, the team will be working through the backlog in manufacturing and e-mails, so we would appreciate your patience.
We thank you for your understanding through this incredibly challenging time and we truly appreciate your messages of support throughout what has been a difficult situation.
Please take care of yourself, your family and we wish all those key workers on the front-line safe passage through the unprecedented circumstances.
Mark Allen
Managing Director

COVID-19: Manufacturing Facility Closed
Dear Valued Customer,
Due to the escalating situation relating to the COVID-19 virus and the recent Government guidelines, the facility will be closed as of 22:00hrs 23 March 2020.
This is not a decision we take lightly, but one made with the well-being and safety of our employee’s paramount in our minds. Obviously, this will have a significant impact on our customers and supply chain management, for which apologies and hope that you understand the reasons for such dramatic action.
During this time, there will be a task force of personnel who will be working remotely to ensure that this suspension in activities is subject to review during the rapidly changing circumstances. We will continue to follow government guidelines and provide regular updates through our website new page.
We thank you for your understanding through this incredibly challenging time and we look forward to resuming our business cooperation when it is safe to do so. Please take care of yourself, your family and we wish all those key workers on the front line safe passage through the unprecedented circumstances.
Mark Allen
Managing Director

CORONAVIRUS (COVID-19): WHAT WE ARE DOING?
Dear Valued Customer,
Orthoplastics Limited takes the growing concerns around the COVID-19 virus very seriously, and is monitoring the outbreak, and its potential impact to our business, very closely. Our primary focus at this time is to ensure the safety of our team members, while ensuring that we continue to deliver the highest quality products to our customers and the patients who rely on them.
At this time, we have not seen any impact to our day-to-day business activities. Orthoplastics has formed a task force whose primary goal is to monitor this outbreak and to ensure that regular and consistent guidance is being provided to our teams. We have designated primary points of contact to ensure that information is being appropriately communicated. In addition, to ensure that our focus on delivering products is not compromised, we have taken the following actions:
Business continuity planning relating to COVID-19 is in place to ensure that we are prepared to handle situations that may arise
Travel guidelines and restrictions are in place
Guidelines have been provided to our teams that cover, among other topics, health and hygiene and additional required facility cleaning, as well as best practices
We are limiting visitors to our facilities
We are encouraging that meetings take place via video or teleconference rather than in person
We have suspended attendance at large conferences and meetings
In addition, we are working closely with our suppliers to ensure that they, too, have implemented appropriate measures to ensure continuity of supply. We appreciate that this is a very fluid matter, with new developments being reported daily. We will continue to monitor this situation with the utmost diligence with the goal of ensuring continuity of supply to our valued customers.
On behalf of all of us at Orthoplastics, thank you for your continued business.

Mark Allen
Managing Director Go Back

New Manufacturing Facility to be Completed by Summer 2020
In September 2019 we announced plans for the opening of an additional manufacturing facility in the UK, it’s been a busy six months for us and we’re excited to update you on the progress of the new building at the Futures Business Park Development in Rossendale.
As with all complex builds, the path to completion has not been a smooth one but we are pleased with the progress we have made and are on target for the facility to be in operation by late summer 2020.
Overcoming challenges of a complex build
As this is not your normal build and there are many complex factors that need to be considered there have been some challenges for us to overcome. One of the biggest challenges we have faced is the size of the hydraulic press and how it will be placed in the building.
The press which is being manufactured in Slovenia weighs 277,300 KGs and will be delivered to us here in Rossendale on 14 articulated vehicles and re-built onsite. Business Development Director at Orthoplastics, Neil Anderton has been overseeing the development and recently visited Slovenia to approve the thermal press for compression moulded sheet material.

Given the size of the press, it will sit in a pit that is the size of an Olympic swimming pool. The planning around this has led to much collaboration between Orthoplastics, our architects and contractors to help us not only overcome immediate challenges but to also foresee future issues and future-proof the building.
A two-phase approach
The building works are being carried out over two stages. The partial completion of the building is on schedule to be complete for the end of February. Once this first stage is complete the press will be delivered from Slovenia and sited in March/April, allowing for the final stage of building to be completed by the end of June.

Following the successful completion of the building, the facility will be ready to begin manufacturing from August and will enable Orthoplastics to further increase its market capabilities with over £5 million of investment annually.
Mark Allen, Managing Director, Orthoplastics said: “This significant investment underlines our belief that there is huge potential for innovation and growth within the Orthopaedic sector. It is also the next step towards achieving our ambition of pioneering improved patient outcomes and the longevity of implantable medical devices.”

Developing our Direct Compression Moulding Capabilities
Orthoplastics has focused on advanced methods of manufacturing UHMWPE and is a World leader in Direct Compression Moulding technology with over 30 years’ experience of orthopaedic plastic manufacturing.
As we strive to improve patient outcomes through innovation, we are excited to announce that we have expanded our direct compression moulding capability with the addition of 5 brand new machines during the last quarter of 2019 to help us advance orthopaedic developments as well as respond to growing market demands and patient expectations.
This decision is part of a long-term business strategy focused on introducing new and improved manufacturing technologies.
Managing Director, Mark Allen said, “We have always been clear that we want Orthoplastics to be known not just as a leading manufacturer and supplier of UHMWPE, but a business that is making recognised contributions to the advancement of the product and its applications across the world.”
Our investment in equipment, technology and in-house capabilities is part of our bigger vision to be at the forefront of setting standards and making new breakthroughs when it comes to manufacturing and designing materials for wide-ranging orthopaedic applications.
Our ultimate aim is to be the best it can be when it comes to innovative UHMWPE design and manufacturing techniques and to ensure that its materials and processes support the manufacture of Orthopaedic limbs and components that can improve the quality of lives for patients through extended product life span, quality and strength.
Creating components with a high gloss finish
Although Direct Compression Moulding is known to be a relatively expensive method of sintering the polymer, it produces components with a high gloss finish.
We have focused on advancing this method of manufacturing UHMWPE to ensure that components produced this way are optimised. We have also pioneered the development of an ‘insert moulding’ process, which allows UHMWPE to be moulded onto or into another steel, titanium or ceramic part.
Medical grade moulding of individual components
Direct Compression Moulding has the capability to produce products which cannot currently be produced, pushing the design envelope for medical grade orthopaedic components. Each component is individually moulded, allowing the finite controlling of all aspects of pressure, temperature and time, during critical consolidation.
By focusing on innovation, this method of manufacture has produced unique over moulding capabilities to produce Mono block components that have a high gloss moulded surface. These design and aesthetically pleasing products provide an alternative market solution to any OEM’s portfolio.
Our Direct Moulding Services
Our Direct Compression Moulding services are at the forefront of new product development. Using our technological expertise, we can bring to market new innovative product solutions to address failure modes typical within orthopaedic implants
Although the technology has been in existence since the 1990's, the new generation of technologies which have been invested in, particularly the 3D printer, allows printed metals and other porous metals technologies to be created.

Orthoplastics Win Two Awards at the Rossendale Business Awards
We are very proud to announce that we have won ‘Business of the Year’ and ‘Employer of the Year’ at the Rossendale Business Awards. This is a great achievement for all of us at Orthoplastics and demonstrates our investment in innovation, our people and our processes to become a Company of Choice.
Organised by Valley at Work, the Rossendale Business Awards is an annual showcase for the achievements, quality and diversity of the local businesses and enterprising people who make Rossendale such a great place to live, work and visit.
This year was the 10th annual Rossendale Business Awards and the ceremony was packed with finalists, sponsors and supporters for a great evening.
There was some tough competition in both categories and we are delighted to have been awarded not only ‘Business of the Year’ but also ‘Employer of the Year’. To win two such highly regarded awards shows that Orthoplastics is a forward-thinking company that has a strong focus on its people.
Our Business Development Director, Neil Anderton attended the awards ceremony on the 4th Dec 2019 at the Riverside in Whitworth and was proud to accept the awards on behalf of everyone at Orthoplastics.
“We see our employees as the business. All employees are aware they can voice their opinions and concerns at any time and they will be heard and actioned. We offer a family atmosphere however work to a corporate standard.
As technology moves forward, we strongly believe investment in our employees is paramount, by identifying the strengths and weakness of our employees we have been able to upskill and redeploy employees to ensure they are working to their strengths.
We feel innovation is at the heart of the future of UK manufacturing, and we continue to provide our customers with the confidence in our long-term investments strategy, whilst demonstrating our strategic partnership and commitment,” Neil Anderton , Business Development Director.
Our recent wins are a great accolade for us and a fantastic way to end 2019. We look forward to next year’s awards ceremony.

A Look Back at 2019 and What’s in Store for 2020
It’s been a busy and exciting year for us here at Orthoplastics and we’d like to thank all our loyal customers for their continued support and take the opportunity to welcome new customers that have come on board.
During 2019 we have achieved many great things and our continued growth has positioned us well for 2020 and beyond. From encouraging females into engineering through employment, work experience and apprenticeship opportunities to continued staff development and collaboration opportunities.
Here are some key highlights of our achievements in 2019:
● We reached 200 employees during the summer and plan to continue recruiting.
● Our Managing Director, Mark Allen had the honour of giving a keynote speech at the 9th Annual UHMWPE Conference.
● 4 Engineering Apprentices joined Orthoplastics and began college courses in September.
● All 39 employees who attended the Highfield Level 2 Award in Health and Safety within the Workplace achieved a Pass, with 3 attendees achieving 100% in the exam.
● We began talks with Lancaster University to explore the potential for setting up a long-term collaborative partnership.
● We began rolling out Lean training across the business to all employees to ensure everyone has a good baseline understanding of dealing with challenges in the leanest manner.
Looking Ahead to 2020
As 2019 draws to a close, we look ahead to 2020 and the exciting developments we have planned. The first quarter of 2020 will see the opening of an additional manufacturing facility in the UK, set to open in February 2020, the new building will enable us to increase our market capabilities with over £5 million of investment annually.
The new facility at the Futures Business Park Development in Rossendale will fast become a centre of excellence for our business and a launch pad for our extensive global growth plans to support plans to be the world-leader in polymeric solutions.
Mark Allen, Managing Director, Orthoplastics said: “This significant investment underlines our belief that there is huge potential for innovation and growth within the Orthopaedic sector. It is also the next step towards achieving our ambition of pioneering improved patient outcomes and the longevity of implantable medical devices.”
The expansion is expected to create around 100 new jobs within the local community and is fully supported by our US parent company, Viant Inc.
Supporting our business expansion, we have also been quality planning ahead for 2020. Our teams have been thinking about all aspects of quality within a new manufacturing area. By forward planning, we are ensuring that our Quality Management System is at the forefront of our plans for Orthoplastics expansion.
We are in the process of validating our Quality management software, by updating our current software to a web-based version. By doing so we are still maintaining all aspects of the quality management system, such as document, audit and corrective action management, but will also enable greater functionality between users, particularly between various sites.
Dates for the Calendar in 2020
We will be attending various events around the world throughout the year. Some key dates for your calendar include:
24 - 28th March - AAOS Annual Meeting in Orlando, FL, USA
9th -11th June - 16th Annual Orthopaedic Manufacturing and Technology Exposition and Conference in Chicago, IL, USA
23rd-26th September - ISTA 2020 Maui in Maui, USA
We’d like to wish all our customers, suppliers and contacts a very Merry Christmas and a prosperous New Year from everyone here at Orthoplastics. We look forward to continuing working with you in 2020.

What’s Been Happening at Orthoplastics in 2019?
It’s been a busy 2019 at Orthoplastics so far, there have been lots of exciting developments, collaboration opportunities and we have continued to focus on employee development. Here are some highlights of what has been happening at Orthoplastics this year.
Lancaster University Visit
Orthoplastics recently spent the day at Lancaster University to explore the potential for setting up a long-term collaborative partnership. The day began with us giving a general overview explaining what it is Orthoplastics does and the services we provide, and where we are heading as an organisation in terms of our future plans and aspirations.
The remainder of the day was spent attending several networking sessions with various departmental leads in order to gauge whether the university can potentially slot in with our organisation’s strategic needs.
The idea was to explore potential projects which will benefit both Orthoplastics and Lancaster University, and we were able to chat with the leads from several faculties including the Data Science Institute, the Engineering Faculty, the Management School, as well as the leads from the Knowledge Transfer Partnerships, Employer Engagement and the lead regarding Student Placements.
The discussions were extremely informative and have allowed us to begin to look into ways in which we can create a mutually beneficial future relationship.
Continued Staff Development
At Orthoplastics, we are committed to ensuring our staff have access to the training they need to further develop their skills and knowledge and enhance their work performance. We are currently rolling out Lean training across the business to 204 employees to ensure everyone has a good baseline understanding of dealing with challenges in the leanest manner.
Our Operational Excellence Manager, Paul Gordon is rolling out the training and with around 40-50% of the team having undertaken the training, he has received some great feedback on the course.
In addition to our Lean training, Team Leader and Customer Services NVQ’s are taking place at co-ordinator and team leader level across the business. We currently have around 10-15 employees undertaking the NVQ’s and will continue to roll them out until all relevant staff have obtained the qualification.
Getting Females into Engineering
Females entering the engineering sector has been a hot topic for the last few years, and here at Orthoplastics we fully support and encourage those who want to work with us or experience what a career in engineering could be like through employment, apprenticeship and work experience opportunities.
We’ve recently welcomed a female Miller to the Orthoplastics team and Lauren Schofield recently completed her apprenticeship with us and is now continuing with her HNC in Engineering. We’re looking forward to welcoming a female student from Burnley College who will be joining us for a work experience placement.
Our work experience and apprenticeship opportunities give those who undertake them a first-hand insight into what a career in engineering involves and can be a great way to introduce engineering to the younger generation.

Quality Planning Ahead for 2020
Orthoplastics development of a new facility planned for 2020, has had the Quality Team thinking about all aspects of Quality within a new manufacturing area. The new building, set to open in February 2020, will enable Orthoplastics to further increase its market capabilities with over £5 million of investment annually.
This is a significant move for the business in a rapidly advancing and innovative market and one that is fully supported by US parent company, Viant Inc. By planning ahead, we are already thinking about our Quality Management system, and how we can utilise this at the new facility.
We are in the process of validating our Quality management software, by updating our current software to a web-based version. By doing so we are still maintaining all aspects of the quality management system, such as document, audit and corrective action management, but will also enable greater functionality between users, particularly between various sites.
By forward planning, we are ensuring that our Quality Management System is at the forefront of our plans for Orthoplastics expansion. Incorporation of SQDC (Safety, Quality Delivery, cost) into manufacturing areas throughout the organisation has enabled manufacturing areas to be at the forefront of collecting quality data and reviewing improvement solutions.
Our Quality team are using data collected from SQDC techniques to refocus our continuous improvement strategy and use of the SQDC tool will align Orthoplastics quality management system and improve communication of quality-related activities to all employees, driving our values into each department.
Achievements of Quality Systems and Accreditations
At Orthoplastics, quality encompasses everything across all aspects of production, from customer service, to design and manufacture of our premium grade medical plastics.
A series of Quality Systems and Accreditations form the foundations of Orthoplastics success. Using the requirements of 21 CFR 820 as a guide, we are certified to ISO 13485 and ISO 14001.
Employing traceability techniques during the manufacturing of all our medical grade product, allows us to regularly assess product quality. All data is recorded and stored for a minimum of 30 years after the manufacturing processes.

9th Annual UHMWPE Conference Update
This time last week we were getting ready to attend the 9th Annual UHMWPE Conference at the historic Union League of Philadelphia. We were also proud sponsors of the event and our Managing Director; Mark Allen had the honour of giving a keynote speech.
Mark spoke about ‘a new novel method for the manufacturing of highly crosslinked UHMWPE using x-ray technology.’ A topic he is passionate about, he spoke of the latest STERIS development of a new method to create high x-rays which allow sterilisation and chemical change in UHMWPE.
With sustainability a hot topic in our industry and across many industries, this sustainable development is deemed the long term solution to Gamma processing using highly radioactive isotopes.
The UHMWPE Conference was a great opportunity for us to learn more about new developments and network with leaders and pioneers in medical grade UHMWPE technology and clinical applications.
View the Keynote Speech
If you weren’t able to attend the conference but would like to view Mark Allen’s keynote speech, please request the presentation by contacting us.

Orthoplastics Sponsor 9th Annual UHMWPE Conference
As the world’s leading producers of UHMWPE, Orthoplastics are pleased to announce we will be attending the 9th Annual UHMWPE Conference on the 10th-11th October 2019 at the historic Union League of Philadelphia.
Not only will we be attending the event, but we are also proud to be sponsoring the event as Bronze Sponsors and our very own Managing Director, Mark Allen will be giving a keynote speech.
The conference brings together engineers, scientists, clinicians, and regulators from academia, industry and FDA to present leading-edge research on advancements in medical grade UHMWPE technology and clinical applications.
The focus of the 9th meeting is on:
- Advances in processing, crosslinking, and sterilisation of UHMWPE
- New methods of processing clinical and retrieval studies of highly crosslinked UHMWPE (HXLPE)
- The performance of thin acetabular liners and knee arthroplasty
- UHMWPE articulations with ceramic and PEEK bearing surfaces
- Vitamin E and new antioxidant technologies for UHMWPE
- Drug delivery from HXLPE
Since our investment in 3 brand new, bespoke UHMWPE (ultra-high molecular weight polyethylene) extruders we have increased our manufacturing capacity by 75% to ensure we are in the best possible position to move with orthopaedic developments as well as responding to growing market demands and patient expectations.
Mark Allen, Managing Director at Orthoplastics has been invited to be a keynote speaker at the conference, where he will be talking about ‘a new novel method for the manufacturing of highly crosslinked UHMWPE using x-ray technology.’
Mark explains more about this latest innovation saying: “STERIS have developed a new method to create high energy x-rays which allow sterilisation and chemical change in UHMWPE. This sustainable development is deemed the long term solution to Gamma processing using highly radioactive isotopes.”
We are looking forward to hearing about the latest developments and innovations at the 9th Annual UHMWPE Conference and hope to see you there.

Further UK business expansion at Orthoplastics
Orthoplastics is pleased to announce plans for the opening of an additional manufacturing facility in the UK.
A world leader in the development and supply of implantable-grade plastics used within the Orthopaedic sector, this expansion is expected to create around 100 new jobs within the local community in years to come.
Official opening expected in 2020
The new building, set to open in February 2020, will enable Orthoplastics to further increase its market capabilities with over £5 million of investment annually. This is a significant move for the business in a rapidly advancing and innovative market and one that is fully supported by US parent company, Viant Inc.

The location of the new facility will be at the Futures Business Park Development in Rossendale (UK). It will fast become a centre of excellence for the business and a launch pad for its extensive global growth plans, designed to ensure Orthoplastics becomes world-leader in polymeric solutions.
Mark Allen, Managing Director, Orthoplastics said: “This significant investment underlines our belief that there is huge potential for innovation and growth within the Orthopaedic sector. It is also the next step towards achieving our ambition of pioneering improved patient outcomes and the longevity of implantable medical devices. Joint replacement procedures such as HIP and knee surgeries are highly successful of course, but we are leading the way in the evolution of new concepts to improve these life changing operations.”
Part of a global, growth strategy
The long-term UK manufacturing investment being made by Orthoplastics demonstrates its confident business strategy. “If the Orthopaedic sector is to grow and thrive, we must embrace new technologies and materials. Our vision and ambition for superior materials solutions perfectly represents how we are putting this strategy into practice with Viant globally.” said Mark Allen.
Providing customers with greater surety
“Innovation is at the heart of the future of UK manufacturing, and this factory will provide our customers with the confidence in our long-term investments strategy, whilst demonstrating our strategic partnership and commitment.” said Mark Allen.
A clear focus on end-to-end solutions
Alton Shader, CEO at Viant commented: “We focus on providing end-to-end solutions for medical device OEMs. Our extensive materials expertise including metals, combined with our experience in engineering, manufacturing, assembly, packaging and sterilization, allows us to bring our customers' medical technology solutions to market.”

Advancing Direct Compression Moulding
As we strive to improve patient outcomes through innovation, we are expanding our direct compression moulding capability with the addition of 5 brand new machines during the last quarter of 2019.
This decision is part of a long-term business strategy focused on introducing new and improved manufacturing technologies. We have always been clear that we want Orthoplastics to be known not just as a leading manufacturer and supplier of UHMWPE, but a business that is making recognised contributions to the advancement of the product and its applications across the world.
Our investment in equipment, technology and in-house capabilities is part of our bigger vision to be at the forefront of setting standards and making new breakthroughs when it comes to manufacturing and designing materials for wide-ranging orthopaedic applications.
Orthoplastics ultimate aim is to be the best it can be when it comes to innovative UHMWPE design and manufacturing techniques, and to ensure that its materials and processes support the manufacture of Orthopaedic limbs and components that can improve the quality of lives for patients through extended product life span, quality, strength etc.
We can create components with a high gloss finish
Although Direct Compression Moulding is a known to be a relatively expensive method of sintering the polymer, it produces components with a high gloss finish. Orthoplastics has focused on advancing this method of manufacturing UHMWPE to ensure that components produced this way are optimised. It has also pioneered the development of an ‘insert moulding’ process, which allows UHMWPE to be moulded onto or into another steel, titanium or ceramic part.

Stuart Green retires after 38 years
Here at Orthoplastics we believe that our staff are the backbone of our business and pivotal to our ongoing growth and success.
It’s with sadness, therefore, that we had to say “goodbye” to Stuart Green, Business Development Director, who officially retired on 29th May after 38 years of loyal service.
Whilst working for Orthoplastics, Stuart has held several senior positions. His knowledge of Orthopaedic plastics, different industries and the latest manufacturing techniques and equipment is unquestionable. As an individual he was highly respected across the medical world.
A key player in the development of our business
Over the years, Stuart has played a big part in the development and growth of our business and we all wish both him and his family well for the future wherever life takes them.
Managing Director, Mark Allen said, “Good luck Stuart. We’ll particularly miss your drive and dedication. Our staff, directors and customers are sad to see you go. I firmly believe that your input spanning more than 3 Decades has helped us to become a world leader in implantable plastics manufacturing and a key player across the medical sector.“
We want to hear from ambitious, talented individuals that want to kick-start their career
Are you interested in joining a progressive, motivated and ambitious team of people that want to pursue an amazing career in a technical environment?
If you think you have what it takes, we’d like to hear from you. Visit our careers page for details of the sorts of jobs we can offer and how you can submit your CV through our skills hub.

A significant component cell expansion
Over the past 12 months Orthoplastics has invested in excess of $4M dollars in a massive expansion to its CNC machining capability.
With the addition of 17 five axis milling centres and 10 lathes, Orthoplastics now has a world-leading component manufacturing cell with 45 state of the art machining centres.
By choice Orthoplastics machining capabilities relate only to plastics, both implant and instrument grade, where we can offer the latest CNC machining centres and lathes. It’s important, therefore, that we keep up-to-date with the latest equipment developments and have the manufacturing capacity to work at optimal levels.
Our investment in new machinery and technology is continuous so we maintain a plant list so customers can appreciate the true extent of our capabilities. Supporting the company’s machinery are Mitutoyo CMM’s and various surface treatments, which are engineered to exceed customer specifications.
We’re so confident in our skilled workforce and industry-leading infrastructure that we believe we can provide high quality components quicker and more cost effectively than any competitor.
Mark Allen, Managing Director, said, “We take great pride in what we do and are committed to providing our customers with a broad range of specialist services. We can only do that if we have the people, technology and equipment in place to respond their requirements. Our ongoing investment programme is extensive and designed to keep us at the heart of our industry in terms of producing UHMWPE implantable materials of a world-leading standard.”
Do you want to find out more about our capabilities? Contact us and send our technical team your questions and we’ll enlighten you.

We’ve increased our UHMWPE manufacturing capacity
Orthoplastics has recently invested in 3 brand new, bespoke UHMWPE (ultra-high molecular weight polyethylene) extruders.
This latest technology significantly increases our manufacturing capacity by 75% ensuring we are in the best possible position to move with orthopaedic developments as well as responding to growing market demands and patient expectations.
Orthoplastics operates a series of high performance ram-extruders. Each has a capacity of 30/tonnes of UHMWPE rods and screw extruders of a 50/tonne capacity.
A primary bearing material for wide-ranging Orthopaedic applications
UHMWPE’s high abrasion resistance and toughness makes it an exceptionally attractive solution for orthopaedic applications due to the many complexities of wear.
Orthoplastics manufacture UHMWPE from Celanese GUR® 1020, 1050 and 1020-E polymer via two distinct processes: (1) ram extrusion, which produces a rod shaped product and (2) compression moulding, resulting in a large sheet of product.
“We are committed to raising standards through investment – the recent addition to our plant of 3 new ram extruders is a part of bigger plan”
We believe our manufacturing systems and equipment are the most advanced and specialised in our industry at the present time. That gives us the confidence to take on even the most demanding of projects knowing that our people, processes and technology are up to the job.

3D printing is the future and we’re leading the way!
Orthoplastics is pleased to add 3D printing to its in-house manufacturing capabilities.
The new Stratasys OBJET 30 machine has the ability to instantly produce design prototypes and fixtures and is very much a new operational tool for modern manufacturing environments.
This recent acquisition has made our intricate design processes and modelling much sharper, quicker and accurate and underpins our core ability to respond to our customers’ complex requirements covering a broad spectrum of orthopaedic designs and applications.
A big performance machine with an amazingly small footprint!
Our decision to invest in the Stratasys OBJET 30 machine was deliberate given it combines the accuracy and versatility of a high-end rapid prototyping machine with the small footprint of a desktop 3D printer.
Orthoplastics comments, “Now that we have a 3D printing capacity, we have the power and tools to create realistic models with specialised properties quickly, efficiently and easily – and all of this in-house to exacting specifications.”
We can create highly realistic 3D models using the latest print technology
“With its astonishingly realistic aesthetics and ability to deliver special properties such as transparency, flexibility and even biocompatibility, we think it represents the future of high performance design in the field of UHMWPE orthopaedic design across the medical world.”

Achieved compliance to ISO 13485 – 2016 Standard
Quality flows through our business and is hugely important to the products, services and support that we give to our customers.
It’s with pleasure, therefore, that we can confirm that have now transitioned to the latest version of ISO 13485:2016.
This has required us to adapt our already robust quality management system and procedures to include a more specific focus on risk management, not only within quality related activities, but areas which feed into the quality management system, such as training and supplier control.
Delivering the highest quality products, consistent with our ethos and overriding medical industry requirements
Orthoplastics ISO 13485 re-certification audit is scheduled for the summer, 2019 and we feel confident that we are ready for this.
Continued compliance to the standard will ensure we can deliver the highest quality products and services to our customers.
Through effective management and maintenance of ISO 13285 across our business, our customers can trust Orthoplastics to be a safe supplier to the medical device industry.
“We remain committed to our customers and take nothing for granted”
We are not complacent about this trust and confidence in our abilities and take nothing for granted. Our team is always looking for ways of ensuring we can move on and provide improved levels of service and product performance.

Advanced Level Apprenticeships
We are pleased to announce that two of our advanced apprentices have graduated with flying colours.
Our congratulations go out to Jake Lord and Timmy Mardy who have now completed their Advanced Level Apprenticeships with us in Engineering Manufacture (Mechanical Manufacturing Engineering) in the Science, Engineering and Manufacture Technologies.
Encouraging our staff to go far
As a business we are committed to encouraging all of our employees to expand their qualifications, and to learn about the latest engineering sciences and advanced manufacturing technologies, to help keep us at the forefront of creating and maintaining standards in our emerging marketplace.


Modern apprenticeships like those our staff have recently completed help us to strengthen our skills infrastructure so that we can compete in the modern world, hence we will continue to encourage schemes of this nature.
In addition to studying at college, both Timmy and Jake have now gained valuable on-the-job experience in their chosen professions as CNC Turner and CNC Miller, respectively.
“We would like to extend our thanks to both apprentices and to congratulate them on their recent results. On behalf of the directors and staff at Orthoplastics, we wish them every success for the future.” (Bernadette Dabbs, Sales and Marketing Director)

Michael Ulanowicz joins the team
Michael Ulanowicz joins the team as Regional Director of Business Development, in USA.
As part of our continued investment in people and customer service systems, I am pleased to announce that Michael Ulanowicz has now joined our team as Regional Director of Business Development (USA), for MedPlast UK.
This new appointment is a sure sign of our commitment to giving our existing customers more, but will also allow us to develop valued global relationships with new businesses and organisations.
Michael will be managing one or more of our strategic healthcare contracts, and challenged with developing lasting partnerships with potential and existing Orthoplastics customers. He will also be responsible for planning, directing and coordinating programs, involving direct contact and negotiations with many of our key accounts.
As and when necessary, Michael will provide engineering support to help promote and grow wider business development opportunities.
With over 26 years experience in plastic manufacturing processes, Michael is well respected across our industry assisting customers on product realization. This has included materials, design for manufacturing and cost reduction. He has specific insight of working with purchasing, quality and engineering departments in numerous orthopedic, dental and cardiovascular companies.
Michael will report directly to Mark Allen, Managing Director of Orthoplastics. “I’m delighted Michael is now part of our valued team, and I’m sure you’ll join me in wishing him every success.”

The importance of our FDA Company Registration
In April 2017, Orthoplastics Ltd became an FDA registered organisation. But why is this important to our business and customers?
Being FDA registered means Orthoplastics comply with the practices of 21 CFR: part 820, which is the code of federal regulations for quality systems relating to medical devices.
A key difference between this and our standard quality management registration is non-compliance to 21 CFR: part 820, which breaks US Law and Regulations.
We have many US-based customers and need the surety that we can meet US market standards and expectations
As a high percentage of our customers are US-based, compliance is paramount for ensuring safe delivery of goods into the US medical device market.
Orthoplastics can provide assistance with FDA product registration as a contract manufacturer within the UK, and our quality team is on hand to provide regulatory and compliance support relating to any new or ongoing applications.
Supporting customers, wherever they are in the world
Mark Allen, Managing Director, said, “We are exceptionally focused on quality as a business. We understand that medical device specifications and products must be consistently delivered to the highest standard. Our various quality registrations prove that we are capable of achieving this and fulfilling the requirements of our customers, wherever they are in the world.”

Orthoplastics passes safety audit with flying colours
Orthoplastics believe that every business has a responsibility to ensure they maintain the highest possible standards of health and safety management at all times.
That’s why we’re exceptionally pleased to confirm that we scored highly in a recent safety audit to OHSAS 18001.
OHSAS 18001 is the International Occupational Health and Safety Management Standard. It provides a framework for the effective management of OH&S, including all aspects of risk management and legal compliance. The Standard addresses occupational health and safety, rather than specific product safety matters.
Our Compliance and Regulatory Leader , Rebecca Field, said: “There are over 90,000 OHSAS 18001 certified organisations across 127 different countries. While we are just one of those, it underpins our belief that all successful and responsible businesses need to adopt best practices when managing the safety of the people that work for them.”
Rebecca added: “In our recent audit, we achieved a pass rate of 80% (to OHSAS 18001) which we are justifiably proud of.”
What benefits has this brought to Orthoplastics?
Apart from our obvious ability to identify hazards and implement preventative controls, it has improved our productivity and general in-house safety culture, it has assisted with pre-qualification processes for our customers, and it has challenged us to be looking for new and better ways of addressing health and safety issues throughout our business.
Our certification to OHSAS 18001 effectively means that we have a clear management structure with defined authority and responsibilities in place, defined objectives for improvement with measurable results, and a structured approach to risk assessment.
“The fact that an independent auditor has visited our site, and thoroughly checked all of our processes and safety management procedures, is a clear indicator to the people that we employ, supply and work with, that we uphold the most stringent and traceable systems possible. It’s also a sign of how important health and safety systems are to our business as a whole.”
Want to know more about our health and safety management certifications and commitment? Give us a call.

A change in ownership for Orthoplastics
A change in ownership for Orthoplastics
It gives us great pleasure to announce that as of January 2017, Orthoplastics (Medplast Group) has been acquired by Water Street Healthcare Partners and JLL Partners.
Both Water Street and JLL are highly respected investment firms with specialist expertise of the healthcare industry. Each business has many years of medical device experience and has committed significant resources to invest in the Company’s future.
Business as usual
While the new ownership will not affect the day to day running or operations of Orthoplastics, it will mean that we have the necessary finance, expertise and advice in place to guide us into the future and ensure that we continue to keep abreast of market demands, technology advances and legalities in the field of orthopaedic UHMWPE innovation.
It’s still the same Orthoplastics
Chris Turner, Quality Manager, said: “Nothing has changed in terms of what we do, or how we do it. It is very much business as usual for us.”
He goes on: “It’s important at this time, that we reassure all of our existing contacts and customers so that they understand there will be no changes to our management structure. Likewise, we will continue to supply the same products and quality services associated with our name, on which we have built our industry reputation and standing.”
So as 2017 starts it seems that, while the ownership of Orthoplastics has changed, its future success and growth in the orthopaedic UHMWPE industry, is even more secure.

More innovation from Orthoplastics
As a world-class leader in the development, supply and manufacture of the highest grade, UHMWPE implantable plastics, we think it’s exceptionally important to push the traditional design limits for orthopaedic components.
Orthoplastics’ investment in Direct Compression Moulding has created exciting opportunities for producing innovative, superior performance products
Direct Compression Moulding
Whilst Direct Compression Moulding has been around since the 1990s, the latest generation of metal 3D printing technology has created new potential for product design. Using Direct Compression Moulding to sinter polymers, Orthoplastics can create products with a high gloss finish, which cannot currently be produced….pushing the design envelope for orthopaedic components.
Investment and innovation are vital
Our investment in Direct Compression Moulding means we now have a unique over moulding capability that produces Mono block components with a high gloss, moulded surface. Given each component is individually moulded, we can control all aspects of pressure, temperature and time, during critical consolidation. This ensures that the finished product’s gloss finish and tolerances are far superior in comparison to machined UHMWPE components.
“It’s important to our business to be at the forefront of developing these specialist parts of our organisation so that we can respond to growing customer and industry expectations. With Direct Compression Moulding, each orthopaedic implant is moulded individually. This allows us to produce a component with a highly polished surface, by removing machining surface definition present on the articulated surface of conventionally machined products. It’s a real breakthrough.”
Mark Allen – Managing Director
This alternative way of producing implant components is being heralded as an important development. It further underlines Orthoplastics’ global commitment to maintaining and setting new standards for high-grade UHMWPE components.
New online presence
Read about our Direct Compression Moulding capabilities on our new dedicated website at www.directcompressionmoulding.com
This explains the capability we now have and about our wider research and development, laboratory and testing services.
Orthoplastics is a Medplast company – science making a material difference.

Neil Anderton fronts development of new implant manufacturing techniques and processing technologies
Implant engineering techniques and manufacturing processes have been through many stages of development over the years.
Neil Anderton, Technical Manager at Orthoplastics, is at the forefront of introducing the market to the latest technologies and advanced, additive manufacturing processes. These are designed to push the boundaries of what is possible in terms of developing high-quality, patient-specific implant solutions.
“Far too many businesses still don’t recognise the incredible potential that additive manufacturing and 3D printing hold”, said Neil. “There is definitely comfort in using more traditional equipment such as CNC technology. Whilst there have been some breakthroughs in CNC tooling and machine software capabilities, this has been limited.”
Neil believes that CNC machines combined with other equipment, such as lasers for cutting, welding or etching, are the way forward. For example, five-axis machining centres can use data from medical scans to create a broader range of patient-specific implant components, especially for skeletal requirements such as the bone, jaw or skull.
A patient-specific approach
To meet challenging OEM requests for patient-specific implant components, Orthoplastics has created a New Products Innovation Group to explore different sciences, control technologies and manufacturing techniques.
In addition to developing custom devices, there are unique composite device constructions that pose real challenges in respect of component fixation and the need for production controls to an exceptionally high tolerance. “We have invested in innovative fixation techniques and specification-controlled environments within our plant, to respond to customer requirements.”
Neil goes on: “Although industry experts generally believe that additive manufacturing will be the death of traditional machining”, I don’t agree.”
Working alongside a leading implant manufacturer, Othoplastics has used net shape and additive manufacturing to produce a manufactured component with tolerances of 0.01 inches, and machined the final tolerances of 0.001 inches required. This involved sophisticated work holdings and machine probing, along with conventional multi-axis machining.
“All of this clearly demonstrates what innovative approaches can achieve in the OEM market”, said Neil.
“Orthoplastics remains committed to this development work and to spearheading different approaches and cutting-edge advancements in processing technologies.”
To read the ODT publication article in full please click here.

Laboratory testing – Orthoplastics lead the way
Without proper testing, validation and certification, new orthopaedic implant products cannot succeed.
Given advancements in technology and increasing regulatory requirements, the need for professional testing, quality management and control during each stage of the production process, has never been more important.
It is widely accepted that compliance, accreditation and associated timelines can be long and expensive when introducing new or improved products. That’s why it’s important to work with a global partner like Orthoplastics that has the appropriate expertise, knowledge and in-house laboratory test facilities. By working with an experienced business, development time (and costs) will be reduced and products will reach the market much quicker.
Investment in technology is key
Orthoplastics is a respected, global expert in the development, production and supply of both implantable grade plastics and biocompatible plastics products for wide-ranging instrument applications. We aim to produce the highest quality, premium grade UHMWPE and work closely with all of our partners to produce new and innovative implant technologies.
Over the years, Orthoplastics has invested time, resources and finance into developing its knowledge of medical plastics, hi-tec manufacturing techniques, test standards and accreditation requirements.
“We have our own fully equipped, laboratory. This is a thriving hub for research and product innovation, ongoing testing and accreditation work. We aim to ensure that each product we manufacture meets the full application, specification and rigorous technical demands required. We believe we provide industry-leading services that are designed and built to support new product developments for our customers.” Neil Anderton – Component Cell Manager
UKAS accredited laboratory
Orthoplastics’ laboratory is UKAS accredited and the majority of projects are carried out in accordance to ISO17025.
Our research and development laboratory has vast experience of testing materials and products manufactured from Ultra High Molecular Weight Polyethylene (UHMWPE), Polyether ether ketone (PEEK) and other polymers, primarily within the medical device sector.
Orthoplastics is a Medplast company – science making a material difference.
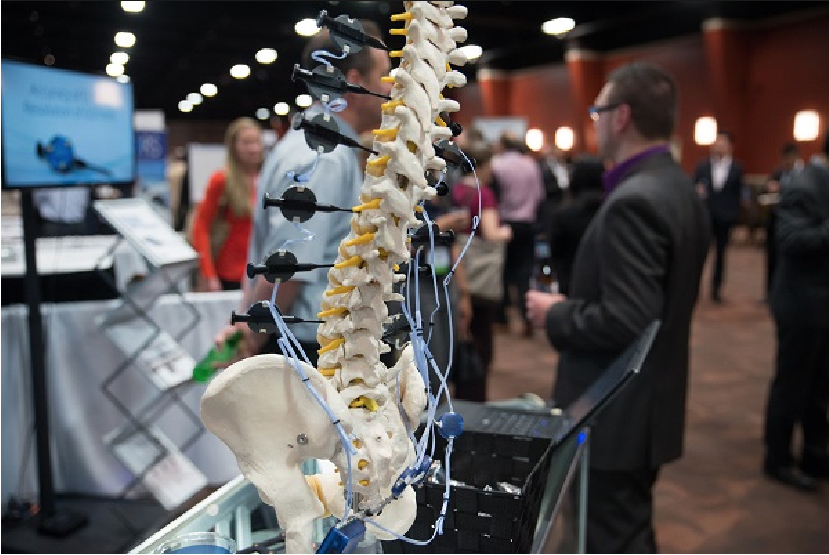
Orthopaedics AAOS and ORS Florida 2016
Orthoplastics attends the global Orthopaedics AAOS and ORS 2016 meetings in Orlando, Florida
(These meetings are a pivotal launch platform for major OEM’s worldwide).
This year we have seen the further growth of UHMWPE material containing protective antioxidant Vitamin E. As the exposed clinical history for these materials is observed, we continue to secure additional patient benefits, including improved long-term performance.
A huge variety of new porous metal products were exhibited at the show. With advancing 3D printing technology, OEM’s introduction of this and the Monoblock component, are key. We have been instrumental in the design and development of OEM’s worldwide. This novel over-moulding technique, combining UHMWPE with a porous metal, removes the need for complex interlocking features within a design.
Orthoplastics has over 30 years’ experience of providing orthopaedic services. We are a recognised expert in CNC machining and offer the following services:
Design Consultation
Prototyping
Pre-Production Validation
Complex CNC Machining
Full Scale Outsourcing.
Our team of skilled technicians use the latest laboratory equipment to provide a range of testing services. These tasks, which are seen by some as complex, demonstrate our proven technical abilities. Our capability in the manufacturing of moulded products, gives us a competitive edge. Reducing product development costs is at the heart of our business, and our teams are currently working together to achieve this goal.
New Orthoplastics’ website goes live
Successfully engaging customers in today’s world requires a Company-wide commitment and recognition that, to stay ahead, a business needs to be constantly reinventing itself.
Orthoplastics, the recognised global leader in UHMWPE machining and manufacturing, with over 30 years’ industry experience to its credit, has recently launched a brand new, browser responsive website for optimal viewing.
“Given the nature of our business and the unique requirements of the medical industry, we have to be continually investing in our machining, testing and development facilities. It’s right, therefore, that our online presence keeps pace with this.
Our new website gives the global market an opportunity to understand our strengths and abilities in the areas of UHMPWE plastics, direct compression moulding, laboratory/test services and component machining. We’re very excited about the launch and see it as a significant step in our development as a business.” Bernadette Dabbs – Sales & Marketing Manager
Complete solutions for orthopaedic components
Over the past 30 years, Orthoplastics has become a leader in the global orthopaedic implant industry.
UHMWPE
Our business supplies and manufactures premium-grade UHMWPE to leading orthopaedic device manufacturers and is a first choice for a range of modern implants/devices.
Read more here
Direct Compression Moulding
A new generation of metal 3D printing technology has created opportunities for innovative product design. We can now produce Mono block components that have a high gloss, moulded surface. Given the nature of production, we have finite control over the final surface finish and tolerances.
Read more here
Laboratory services
Our world-class technicians, well-equipped laboratory and extensive testing and research/development facilities, ensure that new products meet required application, specification and technical demands of the industry.
Read more here
Component machining
By choice, our machining capabilities relate to plastics, both implant grade and instrument. We have an impressive portfolio of high-end CNC machines and lathes that can meet any manufacturing requirement.
Read more here
Orthoplastics is a Medplast company – science making a material difference.

Precision, component machining
Taking new orthopaedic products to market can be a difficult and costly process. The medical industry is tightly regulated and controlled and, therefore, complex to access. Unfortunately many great concepts don’t get past the initial development stage.
The need for quality orthopaedic implant products has grown in recent years and the market is becoming increasingly competitive. With doctors, surgeons, consultants and patients demanding better and more robust treatments, there is now a huge demand for affordable implant innovation.
Professional machining
When developing a product for market, it makes sense to work with a professional machining partner that can provide hands-on help, advice and support, from initial planning and tooling through to materials handling and full-blown production and testing. This will ensure that your investment is secure and costs are controlled.
It takes industry experience, investment in cutting edge equipment and technical insight to produce precision manufactured, orthopaedic products that meet the necessary production, compliance and accreditation requirements set by governing bodies.
Turning concepts into reality
Orthoplastics is a respected, global leader in UHMWPE machining and manufacturing, with over 30 years’ industry experience. We excel in producing the highest grade UHMWPE (Orthopaedic Ultra High Molecular Weight Polyethylene) and work with all of our partners to turn innovative concepts into reality.
Experts in milling, turning, grinding and drilling
By choice, our general machining capabilities relate to plastics, both implant grade and instrument - our CNC machining centres and lathes can respond to any manufacturing requirement.
Alongside our CNC machines, we offer various surface treatments to meet and often exceed customer specifications.
Our component machining services are just part of the story. We provide full consultation, design and engineering and research/development support, as well as rigorous in-house laboratory testing to recognised quality standards.
Over the years our business has been instrumental in producing and testing many new and pioneering orthopaedic implant products that have had a profound effect on patients’ lives.
Our new website is live
Visit our new component machining website at: www.orthoplasticsmachining.com to find out about our 30 years’ experience in the orthopaedic implant industry. This site outlines our component machining strengths and is part of our wider strategy to reinforce Orthoplastics as a world-class leader in UHMWPE machining and manufacturing.

Investment In Quality
Quality is absolutely crucial to our business. Over the years, we have heavily invested in resources and equipment to provide our customers with everything required for regulatory compliance on a global scale.
3rd Mitutoyo scanning heading CMM, along with statistical monitoring software, enable a live data capability analysis. These take traditional inspection and processing to a new level of compliance and control.
Validation and compliance team expansion
As our business grows so must quality. With our recent investments and expansion, we again demonstrate the cost of quality can never be too high, particularly when lives are dependant on our products, services and materials.
The needs of the medical sector are constantly changing to cope with advances in medical science. Orthoplastics continues to respond and to nurture the confidence of its employees. The standards set by the market are increasingly high, but the value added services we provide are designed to match that standard. Most recently, our research centre has realised that their core potential lies in UHMWPE materials hence it is focusing their talents in this area.
Responding to the demands of modern medical science requires a fully integrated approach. At our Orthoplastics facility in Manchester there is evidence of great innovation. This is where our experienced team come together to develop a range of products that are highly efficient and capable.

Expanding Implant Manufacturing Cell
We have been excited by the rapid expansion of our polymer implant-manufacturing cell. The introduction of five state-of-the-art axis milling machines has highlighted our commitment to adopting modern machining practices, leading to shorter lead times and highly competitive pricing.
Component machining is an effective way of converting orthopaedic component concepts into today’s reality. Being world leaders in implantable plastic manufacturing and component machining, Orthoplastics is considered among the leading experts in CNC machining. Our environment-friendly machines and in-house capacity, allow us to provide a full range of machining services.
Some of the services we provide include:
- One-off prototyping
- Pre-production validation runs
- Outsourcing service.
We provide a bespoke design service so we can respond to each customer’s requirement. Rest assured, each and every investment that we make is for our customers and to create even higher standards of performance.
Orthoplastics is committed to providing exclusive product solutions, serving the needs of SMEs. Working with us, gives SMEs a real opportunity to overcome technological, regulatory and financial constraints that can stifle their success. We do this by using our in-house talent and expertise to help with complicated legal requirements such as accreditation, compliance, affiliation navigation processes etc.

UHMWPE Conference Philadelphia 2015
Orthoplastics was pleased to make a presentation at the recent 7th UHMWPE conference in Philadelphia. This brought together experts from various fields such as engineers, scientists and clinicians from academia and industry. It was an opportunity to hear about the latest research, advancements and developments in medical grade UHMWPE technology and clinical applications.
The results of various clinical and retrieval studies of immensely correlated UHMWPE (HXLPE) work was presented to the panellists. Special mention was made about thin acetabular liners and knee arthroplasty performance. In addition to this, the following was also discussed:
- Long terms consequences of 1st generation HXPLEs
- Risks and benefits associated with HXPLE
- A detailed comparison of short term results of anti-oxidant additive stabilized HXLPE and the 1st generation HXLPE
- Thermal processing effects and advancements for HXPLE
- Fibre application advancement of UHMWPE for surgical applications
The organisers accepted three technical research papers and selected two for podium presentations.
The use of high powered x-ray for the manufacture of highly cross-linked UHMWPE, the improved dose uniformity when compared to alternatives such as Gamma and e-Beam
The physical/mechanical performance of highly cross-linked UHMWPE processed using the new novel X-Ray facility.
The use of highly modified UHMWPE in spinal applications, when compared to the costly modern day spinal biomaterials.
Major Contract Win
In October 2015, Orthoplastics secured a major machining contract win with one of the World's leading orthopeadic device manufacturers. This prestigous and exciting development has resulted in the acquisition of three new 5 axis CNC machines that will increase the overall capacity within the machining department.
As a world leader in implantable plastic manufacturing and component machining Orthoplastics have the expertise to navigate customers through the complexities of production, compliance and accreditation, getting medical component products to market quickly and saving customers valuable time and money.
If you would like to know more about these unique services then click here

Orthoplastics China Expansion
The expansion of Orthoplastics China has taken place over many years. Kelvin Wu has represented us in this territory and has been instrumental in securing our success. As part of our planned growth in this region and within Asia generally, Mr Wu has now been appointed as our official representative for China, Japan and Korea.
In addition to Kelvin’s new geographic responsibilities, we have expanded the role of Bernadette Dabbs to include sales, marketing and account management, in addition to her already successful leadership of our customer services team.
Orthoplastics is solely dedicated to the medical sector around the globe. There are no geographic boundaries when it comes to helping our customers. This is part of the strategic growth and expansion strategy.
We firmly believe that quality standards are maintained throughout the world because of our certification to ISO 9001, ISO 13485 and ISO 14001. The supply chain mechanism and distribution network of Orthoplastics, ensures that products can be transported to each corner of the globe, in full compliance with relevant security and safety standards. The export quality of our products is unrivalled - we are considered to be the market leaders in providing specialist guidance and support to customers in the medical sector.
The growth strategy of Orthoplastics is evidence of great leadership and rigorous quality assured products and services, ranging from cross-linked UHMWPE and Vitamin E UHMWPE, component machining, to direct compression moulding and laboratory services.

World Class Test Laboratory
We adopt the most stringent of orthopaedic test methods and global standards. Over the years, we have developed a world-class testing facility, compliant with ISO 17025. To further expand this capacity and requirements from OEM customers, we are now offering long-term device package testing.
The validation and characterisation of polyethylene often crosses the boundaries into packaging, especially when it comes to long-term shelf stability.
Our laboratory is fully equipped with:
Nicolet FTIR
Netzsch DSC
Cambridge Polymer Group Swell Ratio Tester SRT-1
Multi-axis Tensile Test
Oxygen bomb.
Our lab is equipped for multi-tasking hence we are able to deal with a number of jobs consecutively. With our extensive range of testing equipment, ultra-high molecular weight polyethylene and other polymers have become key products.
We have invested in the latest technological equipment so that we can innovate different ideas and products. We conduct related research and development in our laboratory so that we can respond to market trends and are more able to serve our customers. The whole concept of the implantable industry is founded on the rigorous testing and manufacturing of plastics products in the laboratory, hence we are extremely proud of our achievements.