
Laboratory testing – Orthoplastics lead the way
Without proper testing, validation and certification, new orthopaedic implant products cannot succeed.
Given advancements in technology and increasing regulatory requirements, the need for professional testing, quality management and control during...
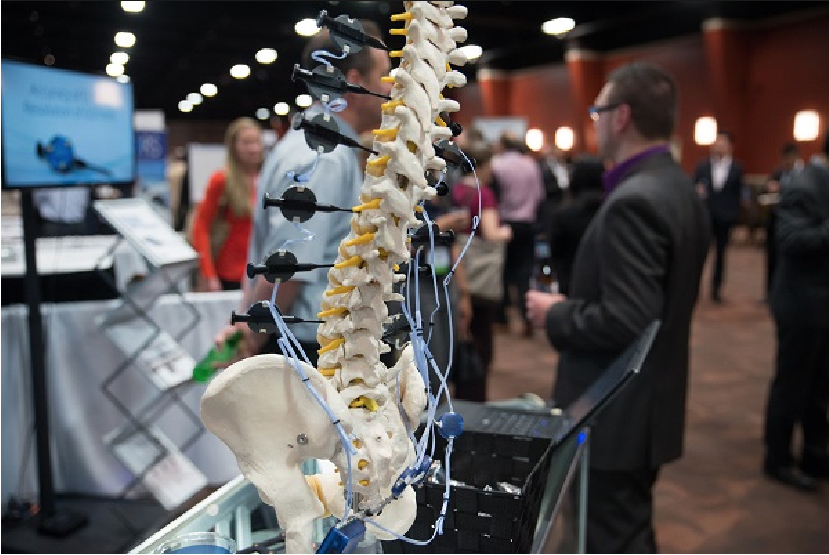
Orthopaedics AAOS and ORS Florida 2016
Orthoplastics attends the global Orthopaedics AAOS and ORS 2016 meetings in Orlando, Florida
(These meetings are a pivotal launch platform for major OEM’s worldwide).
This year we have seen the further growth of UHMWPE material containing...
New Orthoplastics’ website goes live
Successfully engaging customers in today’s world requires a Company-wide commitment and recognition that, to stay ahead, a business needs to be constantly reinventing itself.
Orthoplastics, the recognised global leader in UHMWPE machining and...

Precision, component machining
Taking new orthopaedic products to market can be a difficult and costly process. The medical industry is tightly regulated and controlled and, therefore, complex to access. Unfortunately many great concepts don’t get past the initial development...
Read More
Investment In Quality
Quality is absolutely crucial to our business. Over the years, we have heavily invested in resources and equipment to provide our customers with everything required for regulatory compliance on a global scale.
3rd Mitutoyo scanning heading CMM,...